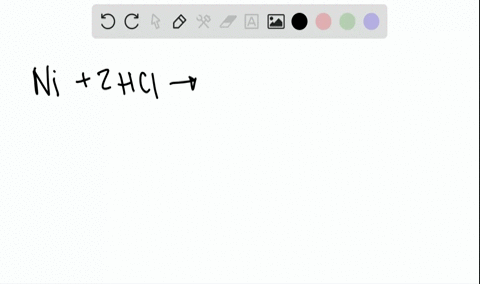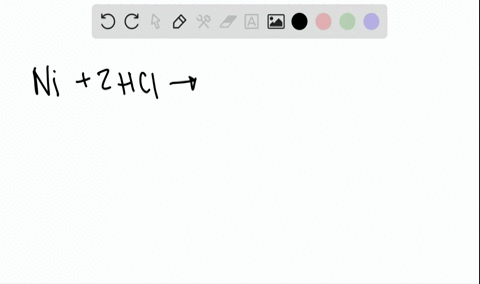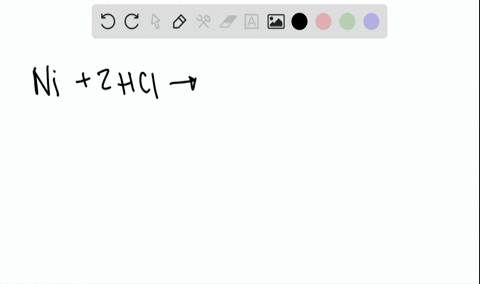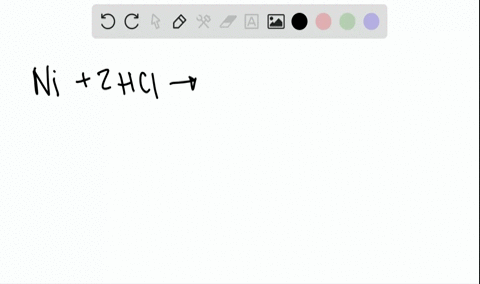
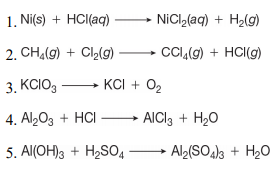
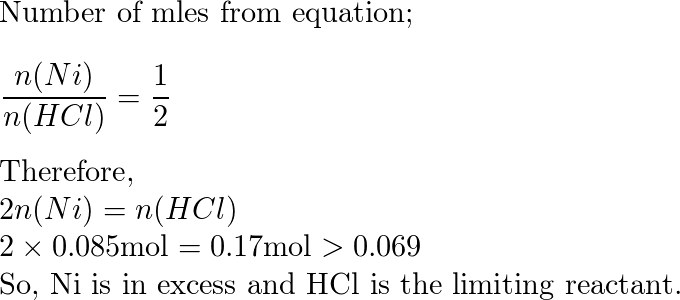Two-Electron HCl to H2 Photocycle Promoted by Ni(II) Polypyridyl Halide Complexes | Journal of the American Chemical Society

Answer in one sentence. Predict the product of the following reaction. Nitrobenzene→Sn/conc⋅HCl? - Chemistry | Shaalaa.com

Effect of HCl concentration on desorption of Ni(II) from DHHC (amount... | Download Scientific Diagram

Cr/Co/Cu/Fe/ Mn/Ni, HCL, Lumina, NO TIMER, 4-pin, Coded - Cableless, PerkinElmer N3050217 compatible

a) Draw the mechanism for the reaction of tin/HCl with m-nitroacetophenone. b) Why is sodium hydroxide added to the reaction? What tin compounds are produced? How do you separate the tin salts

Electrochemical Preparation and Post-treatment of Composite Porous Foam NiZn Alloy Electrodes with High Activity for Hydrogen Evolution | Scientific Reports